For US Healthcare Professionals
I am a
Rheumatology Specialist
For US Healthcare Professionals
Full Prescribing InformationTREMFYA® is the only* dual-acting† IL-23 designed to neutralize inflammation at its cellular source1
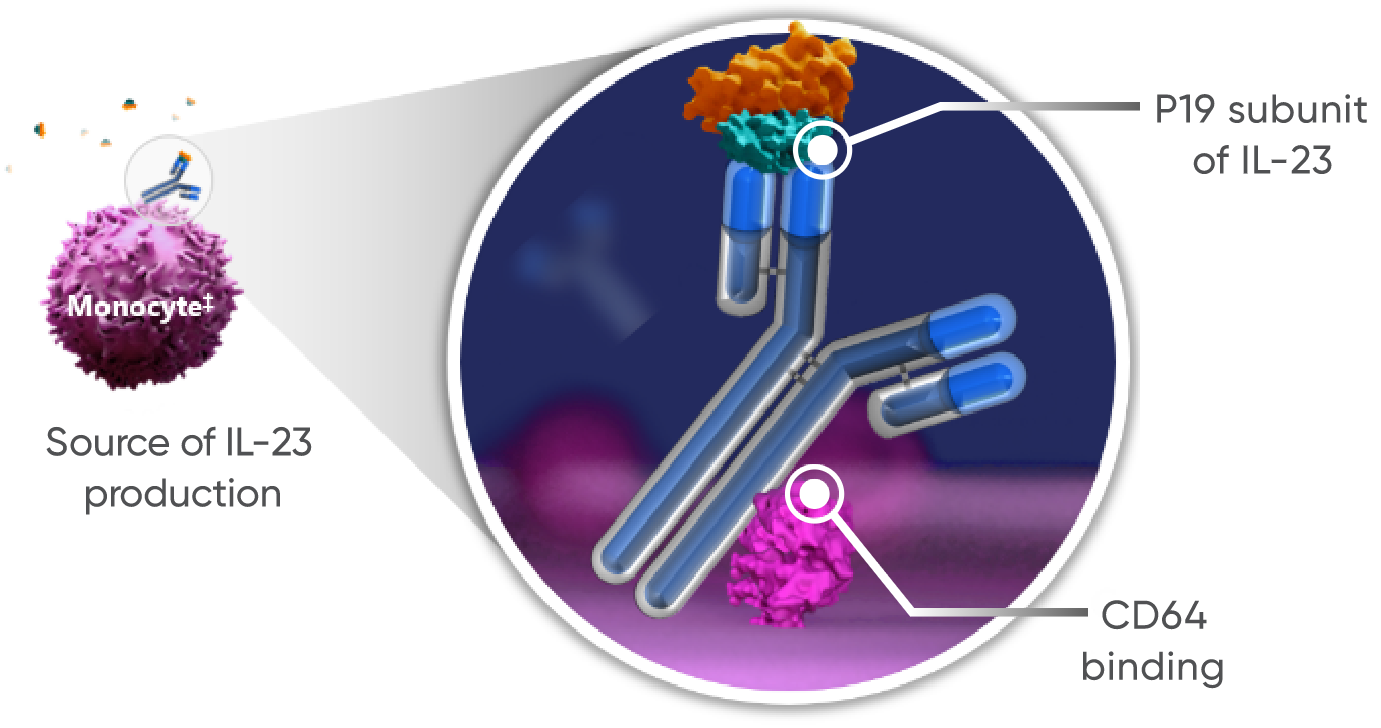
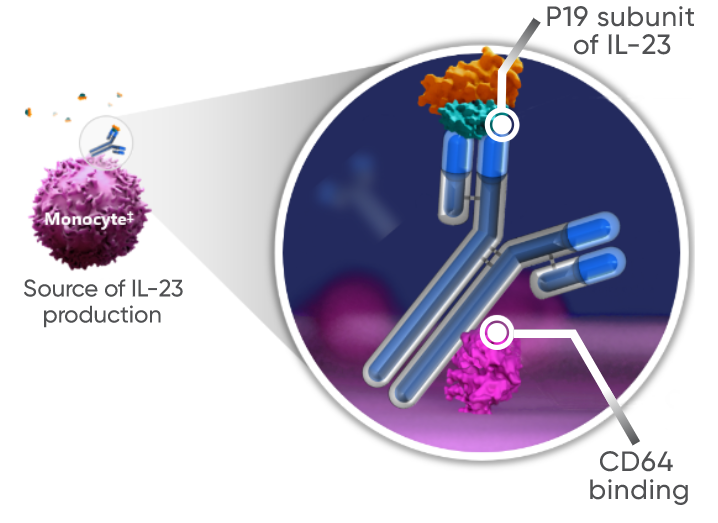
‡In vitro studies were
conducted using an
inflammatory monocyte
model.
Findings for dual-acting are limited to in vitro studies that demonstrated
guselkumab’s binding to CD64 in an inflammatory monocyte model.
*”Only” based on approved IL-23 inhibitors for active PsA as of April 2024.
†TREMFYA® works by:
- Blocking IL-23, a cytokine responsible for inflammation
- Binding to CD64, a receptor on cells that produce IL-23
The clinical significance of these findings is unknown.
CD64=FcyRI; IL=interleukin.
Reference: 1. Krueger J, Eyerich K, Greving C, et al. Poster #LB989. Differentiation of therapeutic antibodies targeting IL-23. Presented at the 2022 Society for Investigative Dermatology.
TREMFYA® blocks IL-23 and regulates key pro-inflammatory cytokines
- TREMFYA® is the only fully human IL-23i1
- TREMFYA® has been shown to decrease the elevated baseline serum levels of IL-17A, IL-17F, and IL-22 in patients without fully blocking IL-17A/F1,2*
The clinical significance of these findings is not known.
IL=interleukin.
*In evaluated subjects with PsA, serum levels of acute phase proteins C-reactive protein, serum amyloid A and IL-6, and
Th17 effector cytokines IL-17A, IL-17F, and IL-22 were
evaluated at baseline. Serum levels of these proteins
measured at Week 4 and Week 24 were decreased
compared to baseline following guselkumab treatment. The relationship between these pharmacodynamic markers and the mechanism(s) by which guselkumab exerts its clinical effects is unknown.
References: 1. TREMFYA® (guselkumab) [Prescribing
Information]. Horsham, PA: Janssen Biotech, Inc. 2. Seibert S, Coates LC, Schett G, et al. Modulation of interleukin-23 signaling with guselkumab in biologic-naive patients versus tumor necrosis factor inhibitor-inadequate responders with active psoriatic arthritis. Arthritis & Rheumatology. 2024;76(6):894-904.